Publication Details
Authors: Lianzijun Wang, Zhongqiang Qiu, Myeongwoo Lee
Journal: microPublication Biology
Year: 2021
DOI: 10.17912/micropub.biology.000485
PMID: 34723150
PMC: PMC8553547
Abstract
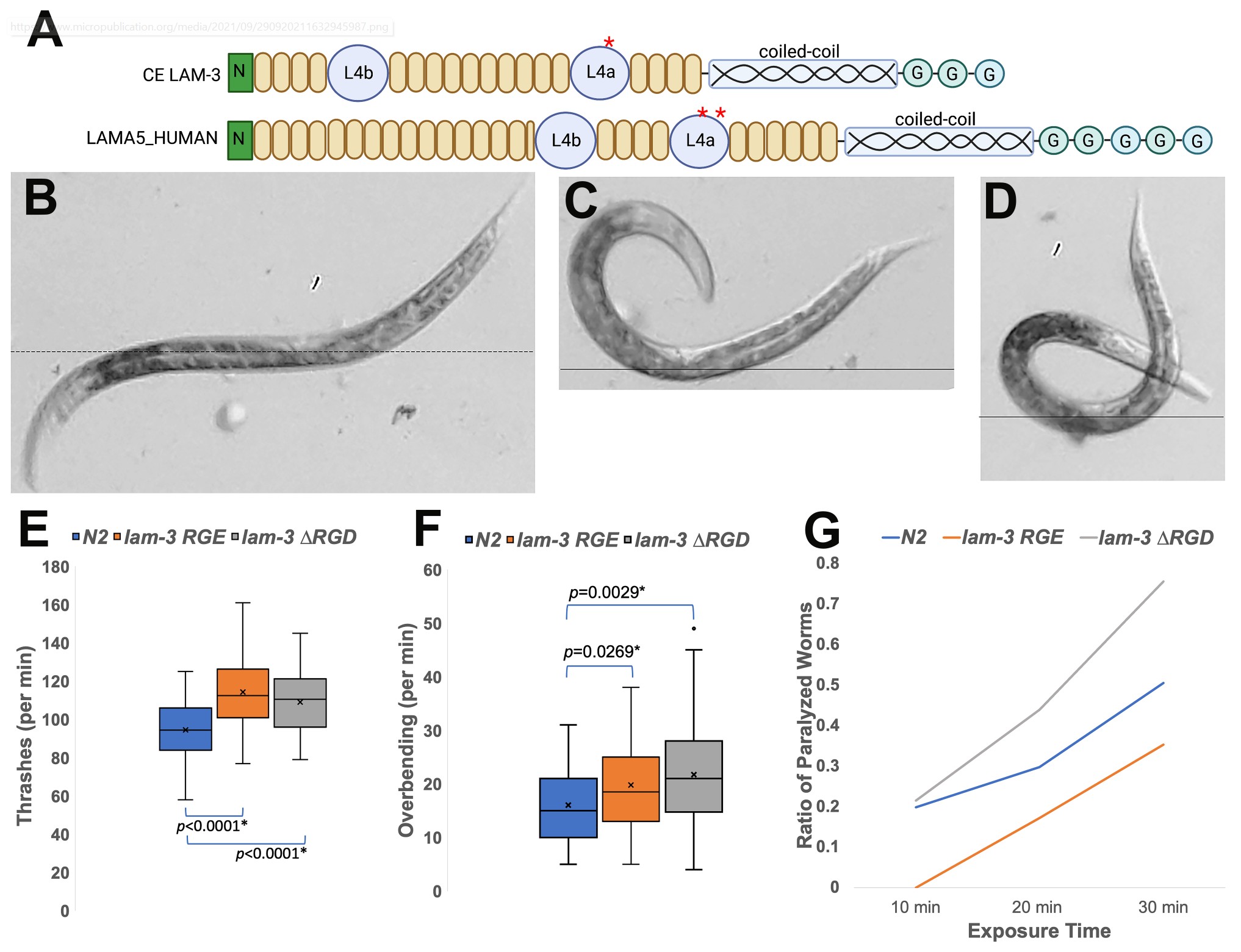
The amino acid sequence Arg-Gly-Asp (RGD) is a cell-binding motif for extracellular matrix proteins. Initially found in fibronectin, the RGD motif is also found in LAM-3/laminin α chain in C. elegans. Laminin, a heterotrimeric glycoprotein, is a major component of the basement membrane. Mutations in laminin subunits disrupt the extracellular matrix and inhibit cell adhesion. This study characterizes the RGD motif in lam-3/laminin α. Two mutations, lam-3 RGE and lam-3 ΔRGD, were generated. The mutants revealed that the RGD motif participates in motility regulation, indicating that cell-laminin interaction contributes to body contraction control.
Keywords
laminin α, RGD motif, extracellular matrix, C. elegans, motility
Citation
Wang, L., Qiu, Z., & Lee, M. (2021). Mutations in the cell-binding motif of lam-3/laminin α reveal hypercontraction behavior and defective sensitivity to levamisole in Caenorhabditis elegans. microPublication Biology. https://doi.org/10.17912/micropub.biology.000485